This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which sections of the website you find most interesting and useful.
Medical Device Regulation (MDR): role local EC + timelines EC
If the clinical investigation with a CE-marked medical device, that is used within its intended purpose, is not considered to be a PMCF, only positive advice from the relevant EC(s) is necessary and “Regulatory pathway: EC only” needs to be followed. The “Other clinical investigations” are regulated under the Royal Decree of 18 May 2021 and as such a timeline of 45 days is applicable to issue an EC-decision.
We refer to the pathway in the scheme below:
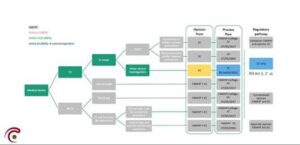
Also for Other clinical investigations involving ‘in-house’ devices or custom-made devices of which data will not be used for conformity assessment, a timeline of 45 days is applicable to issue an EC-decision (Regulatory pathway: Separate opinion FAMHP and EC).
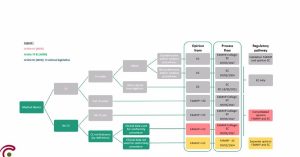
In the Royal Decree of 18 May 2021, there is mentioned in article 36 for multicentric studies:
According to KB 21 May 2021 (medical devices)
- T0: Submission at central and local EC
- T0: Central EC requests local advice on 1° and 2° (cf. article 36)
- T5: Advice of local EC to central EC (accept or decline, except 2°)
- T45: Definitive advice to applicant and local EC
When a study falls under the Belgian law on experiments (law of 7 May 2004), a local EC has 25 days to forward an opinion to the Central EC, not 5 days as currently stated in the Royal Decree of 18 May 2021 for other clinical investigations.
We thus see a discrepancy between the law on experiments (law of 7 May 2004) and the Royal Decree of 18 May 2021 regarding the timelines. We also notice a discrepancy in giving advice about the specific documents. A local EC should also consider “the adequacy and completeness of the written information to be provided and the procedure for obtaining consent as well as the justification for research involving persons who are unable to give consent or whose consent cannot be obtained because of the urgency as to their participation in an experiment;”(d.i. 1° Bijlage I, d) 4 ) and should not only give advice about 2° .
We propose to align the timelines between the different legal texts and to give local ECs 25 days to provide their advice to the central EC, as well as give the local EC the possibility to give advice about the ICF.
